Chemists first took a serious interest in 9-anthraldehyde in the late 19th century, drawn by the unique way it bridges organic functionality with aromatic stability. Stemming from anthracene, a hydrocarbon derived from coal tar, 9-anthraldehyde has always been linked to both industrial progress and academic curiosity. Researchers realized its aldehyde group, sitting neatly at the 9-position, opened doors to endless derivatization. Over the decades, scientists pushed deeper into its potential, especially as modern organic synthesis advanced. Today, people look at 9-anthraldehyde with the same fascination that drove early coal chemists, except now there's a clearer sense of the opportunities—and risks—this compound brings.
9-Anthraldehyde stands out among anthracene derivatives. It carries both the rigidity of the anthracene ring and the reactive edge of the formyl group. Most often, it arrives as an off-white to pale yellow crystalline powder, easy to pick out from a stash of aromatic compounds by the subtle color and distinctive odor. The molecule offers more than novelty; it carries significant potential in building blocks for dyes, pharmaceuticals, and even advanced materials science. In the lab, it often finds its way into custom syntheses simply for the flexibility and accessibility it provides.
The physical features of 9-anthraldehyde make it easy to handle for a practiced chemist. Its melting point usually runs in the 100–110°C range. Chemically, it acts as any decent aromatic aldehyde would: reactive at the carbonyl carbon yet shielded by the bulk of the anthracene backbone. Its solubility drops off in water but improves in most organic solvents, which shows its nonpolar heritage. In the lab, you feel its stability—a bonus for storage and purity. But the aldehyde group will not sit idle. It attracts nucleophiles and participates eagerly in condensation and addition reactions.
In practical settings, the purity of 9-anthraldehyde matters, especially for research or pharma use. Chemists usually look for a product above 97 percent purity, limiting contaminants from related aromatic compounds. Labeling stresses hazard warnings around skin and eye contact, underlining the care needed, especially for those unaccustomed to handling aromatic aldehydes. Batch numbers, manufacturing dates, and storage advice show up on most containers, all underscoring how detail-driven the safe use of this chemical has become.
Anthracene’s conversion to 9-anthraldehyde rarely follows a single strict route. Most syntheses rely on selective formylation at the 9-position, often through a Vilsmeier-Haack reaction. In this classic approach, anthracene meets a mix of POCl3 and DMF, generating the reactive intermediates needed for gentle formylation. Sometimes, chemical innovators tweak conditions, using less aggressive reagents or protecting groups to drive selectivity. Each method trades off yield, purity, reagent cost, and waste. With rising interest in greener chemistry, labs have moved to optimize solvent use and limit toxic byproducts, but even with modern tricks, producing high-quality 9-anthraldehyde takes experience and tight process monitoring.
The real power of 9-anthraldehyde kicks in during downstream reactions. The aldehyde group makes it perfect for Schiff base formation, giving new imines for coordination chemistry and materials research. The compound also serves as a starter for cyclization, Grignard additions, and various condensation reactions. With a crowded aromatic system, 9-anthraldehyde sometimes surprises by holding together through harsh conditions, letting chemists explore reactivity and stability in tandem. Each modification or derivatization extends the molecule’s reach—new dyes, ligands, or intermediates for pharma or electronics. Over several years in the lab, I’ve found 9-anthraldehyde one of those compounds that rarely fails to deliver a predictable result, so long as you respect its quirks and moisture sensitivity.
Historically, people have known 9-anthraldehyde by the systematic name 9-anthracenecarboxaldehyde, and it appears in chemical catalogs under both names. Literature sometimes refers to it as anthracene-9-carbaldehyde or just anthraldehyde for short. As a specialty chemical, it usually shows up under the chemical abstracts number, which helps distinguish it quickly during ordering or source comparison. This jumble of names can throw off students or researchers new to the field, but a seasoned organic chemist learns early to check the structure rather than trust the label.
Handling 9-anthraldehyde requires awareness and respect. Although not notorious for acute toxicity, it brings the same health risks common to aromatic aldehydes. Prolonged inhalation or contact leads to respiratory discomfort, skin irritation, or ocular issues. Every experienced chemist knows to use gloves and eye protection. Fume hoods are a must, not just a suggestion. Waste disposal raises its own problems, given the persistence of anthracene metabolites in the environment. Increasingly, regulations call for tighter logging of use and disposal, especially in research and pilot plant settings. From a training perspective, regular refresher courses on chemical hygiene and proper spill response have become standard practice wherever 9-anthraldehyde gets handled.
People have always valued 9-anthraldehyde most for its flexibility in synthesis. Its role as an intermediate in dye production, especially for anthraquinone derivatives, built a bridge to the textile and pigment industries. In recent years, its capacity to form functionalized ligands for metal-organic frameworks, sensors, and advanced materials expanded well beyond its old industrial roots. At research universities, it serves as a classic starting point for methodology development in organic and inorganic chemistry. Some pharmaceutical development teams explore it for flavonoid synthesis and custom aromatic frameworks. As analytical techniques evolve, the compound gets pulled into fluorescence studies or sensor design, thanks to the robust light absorption and emission properties of the anthracene ring.
The evolution of research around 9-anthraldehyde reflects the broader trends in chemical sciences. Early studies honed in on synthetic routes and characterization. Chemists now look at greener processes, catalysis, and tandem reaction schemes to expand what 9-anthraldehyde can do with less waste. Material scientists experiment with its structure for organic electronics, seeking to extend conjugation or impart new electronic features through careful substitution. In academic labs, it remains a useful substrate for teaching fundamental reactions involving aromatic aldehydes and for exploring structure-activity relationships in ligands or fluorescent probes. Each year, a steady trickle of new studies keeps the field lively, with innovation springing not just from the molecule itself but from the way researchers combine it with modern techniques and technologies.
Although the acute danger from 9-anthraldehyde doesn’t rise to the level of more notorious aromatic compounds, chronic exposure raises real environmental and health questions. Cancer research involving anthracene derivatives shows mixed findings; the formyl substitution appears to alter bioavailability and metabolic fate. Toxicologists push for more data on breakdown products, worried about persistence in soil and water, especially with expanded industrial or research use. As regulations tighten, research groups focus on exposure limits and look closely at metabolic pathways in model organisms. Substitution of safer reagents or engineered controls for process containment forms the logical next step in minimizing occupational risk.
The trajectory for 9-anthraldehyde will likely bend toward applications in smart materials, organic electronics, and sustainable chemistry. Newer green chemistry protocols, whether by solvent replacement or improved formylation catalysts, promise cleaner processes and broader access. With molecular design tools improving, modifying the anthracene framework using the aldehyde functional group looks increasingly attractive for chemists building next-generation molecular switches or light-reactive systems. Toxicology will continue to shape how and where 9-anthraldehyde finds its uses—industry and academia must work together to chart these new paths responsibly, matching innovation with stewardship.
Not every molecule with a complicated name sits on a dusty shelf in a forgotten lab. Take 9-Anthraldehyde, for example. Chemists may call it an aromatic aldehyde, but its value doesn’t end at the molecular level. I spent some time around research teams focused on organic synthesis, so I’ve seen firsthand how 9-Anthraldehyde finds its way into real science, and why those applications actually matter.
The world wants brilliant colors and high-performance materials, and anthracene-derived compounds often step in to fill the gap. 9-Anthraldehyde serves as a stepping stone for these products. Dye manufacturers often select it as a building block, especially when seeking intense hues for fluorescing dyes. Universities and tech startups use those dyes to light up advanced sensors, DNA sequencing experiments, and even security tags you can’t spot with the naked eye.
A small molecule such as 9-Anthraldehyde helps kickstart these high-impact projects. I once visited a startup using anthracene-based dyes to create ink that changes color under UV light. They designed this for secure documents and event tickets, combatting counterfeiting in a way that felt both practical and futuristic. Without 9-Anthraldehyde in that supply chain, that ink never would have come to life.
New pharmaceuticals demand starting materials that bring both versatility and the right chemical backbone. 9-Anthraldehyde acts as a key intermediate, not only because of its reactivity, but because drug development often pushes for complex, multi-ringed systems. This tiny piece of chemistry opens the door to molecules with anti-tumor or antibacterial potential, depending on how researchers shape it.
Working in academic labs, I saw researchers attempt hundreds of reactions, tweaking molecular fragments until they got something promising. 9-Anthraldehyde stood out for its stability on the bench and its ability to react when called upon. Not every experiment pans out, but having a toolbox that includes reliable starting materials speeds up both successes and learning from failures.
People seldom talk about how something gets from the lab to the market. 9-Anthraldehyde production generates hazardous by-products, which presents a safety challenge. Occupational exposure can irritate the skin and eyes — something chemists watch for closely. Regulatory agencies, like OSHA and the European Chemicals Agency, have highlighted the need for strict controls in handling and disposal.
Manufacturers can run closed systems and invest in high-quality ventilation to limit airborne exposure. Regular monitoring, along with the right training, goes a long way in creating safe production environments. Researchers are already designing greener synthesis routes, with less hazardous waste and reduced worker risk. These efforts tie together scientific freedom with accountability to people and the environment.
Basic chemicals like 9-Anthraldehyde rarely make news headlines, yet the world leans on these unsung molecules to fuel innovation. Whether you see its influence in a glowing security label, next-generation medicine, or a new high-tech sensor, the reason for its presence traces back to a combination of reliability, reactivity, and longstanding scientific curiosity. Keeping an eye on safer processes while harnessing useful chemistry reflects lessons learned over decades — and keeps doors open for what’s next.
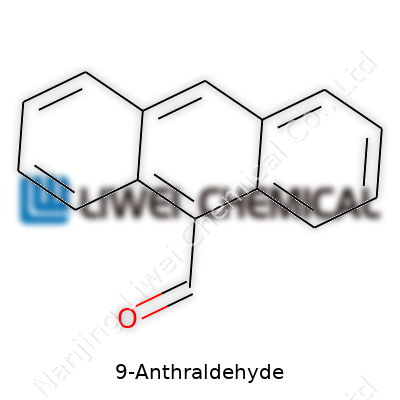
Not every compound in a chemistry lab sparks curiosity, but 9-Anthraldehyde gets attention for a reason. The chemical formula for 9-Anthraldehyde is C15H10O. A single look at its formula and structure shows why it matters, especially for anyone dabbling in organic synthesis, pigment production, or even the development of new pharmaceuticals. My first time handling anthracene derivatives I remember the pungent smell and the deep, orange-red hue of crystals caught under lab light. That was the first reminder that organic molecules can be both relentless and beautiful.
Organic chemistry never stood still. Anthracene, the backbone of 9-Anthraldehyde, pops up in both textbooks and patent documents for a reason. Add an aldehyde group at the familiar "9" position, and the character of the molecule shifts. This new function serves as a handle for countless reactions, turning a simple three-ring hydrocarbon into a doorway for synthetic possibilities. I’ve met researchers who used it as a bridge to make bigger, more elaborate ring systems. Some squeeze out new dyes or fluorescent markers, while others look for pharmaceutical leads, hoping something with this framework binds uniquely to a biological target.
Every time a new compound appears, practical questions follow. Can this molecule help in making better solar cells? Does it build more stable dyes? For 9-Anthraldehyde, those possibilities are not just idle speculation. Its rigidity, derived from the fused aromatic rings of anthracene, works well for forming organic semiconductors. Chemists use it as an intermediate, trying to control light absorption or tailor the emission of organic LEDs. At one conference, someone remarked that molecules like this quietly fuel advances in energy technology without getting headlines.
Taking one look at 9-Anthraldehyde’s structure, I remember my first run-in with its reactivity. Aldehydes demand respect. That carbonyl group works hard in a flask, often requiring ventilation and close supervision to avoid unwanted surprises. There’s also a reality check: organic compounds with aromatic rings, especially those derived from coal tar routes, raise environmental questions. Anthracene itself often comes from such sources, which keeps sustainability on my mind. Cleaner synthesis through renewable starting materials has always been the golden ticket, even if not every lab can afford it. I've seen green chemistry startups try to answer these challenges, aiming for creative routes that spare both resources and researchers.
My own experience in the lab taught me that having the right building blocks like 9-Anthraldehyde sets the pace for breakthroughs. Students often ask whether the exact position of an aldehyde matters, and the answer is always yes. Positions on the aromatic ring control not only reactivity but also how a compound behaves under a UV lamp or inside a living cell. As chemists, we chase these nuances relentlessly. Recognizing that, at the heart, it’s still about piecing together molecules, testing their properties, and figuring out how they can make life just a bit brighter, cleaner, or healthier.
Working with chemicals often seems routine to anyone who's spent time in a lab. Bottles and powders fill every shelf, with warnings printed on labels that can start to fade into the background. 9-Anthraldehyde slips into this landscape almost quietly. It's a chemical found in many organic synthesis projects, especially across research in dyes, photoinitiators, and pharmaceuticals. Yet, familiarity doesn't erase real risk.
As someone who's handled fine chemicals, I always remember the way gloves pick up that faint, sticky scent of aldehydes. 9-Anthraldehyde, with its yellowish hue, gives off a faint odor, a subtle warning signal. Direct skin contact can lead to irritation. Eyes don't fare any better — even a small amount can cause a burning sensation, watering, and redness. Inhalation isn't a favor either; the dust acts fast, setting off sneezing or coughing, sometimes leading to more serious lung symptoms if exposure is prolonged.
There's another risk that sits in wait during any transfer or weighing: fine powder drifting into the air. Many seasoned chemists know the frustration of cleaning up after a spill, but with this compound, every stray grain could mean a risk of inhalation or accidental ingestion. Following institutional safety data, many universities recommend gloves, goggles, and dust masks at all times, even if only a few milligrams are needed.
Many institutions and regulatory bodies, such as the European Chemicals Agency, have flagged 9-Anthraldehyde as hazardous because of its impact on skin and eyes. More than one safety advisory links its aldehyde group to toxicity concerns. Breathing in powders isn’t something people consider unless prompted by a sour whiff or an accidental cough. Chronic exposure is another layer, with potential long-term health effects that still aren’t fully understood, mostly because comprehensive toxicity studies are limited.
Plenty of accidents don't stem from ignorance, but from comfort bordering on complacency. I remember once watching an experienced colleague skip the dust mask out of habit, convinced he had “done this hundreds of times.” That overconfidence led to a coughing spell and a dash for the eye wash. Since then, nobody in that lab has gone back to handling 9-Anthraldehyde bare-faced. Complacency in the lab grows quietly, pushed along by routine and familiarity, making chemicals like this especially tricky.
Culture shifts in science come from individual habits. Encouraging accountability solves hazards more than any posted sign. Supervisors who practice what they preach, peer reminders without judgment, and well-maintained safety gear at every bench all help. Access to up-to-date safety data offers a strong backup, so everyone knows what they're up against, not just what the last person said.
The hazard from 9-Anthraldehyde isn't a myth born from regulatory red tape. It's as real as the burn that comes after rinsing skin or eyes, as present as the feeling in your lungs after a careless sniff. Anyone handling it should think of gloves, goggles, masks, and a fresh respect for chemical hazards as part of their responsibility to themselves and their colleagues. Each choice ripples out, shaping safer labs for new generations of researchers. Safety isn’t paranoia—it's part of the job.
In the world of aromatic aldehydes, 9-anthraldehyde rarely shows up in conversations outside a research lab. Yet, for many working with organic electronics or dyes, knowing details about this compound, like its melting point—reported consistently at about 78-79°C—can impact more than just a day at the bench.
Years ago, I watched a colleague struggle to synthesize clean anthracene derivatives. The project felt frustrating, not because the reactions failed, but because purification kept stalling right around the melting point. Every organic chemist remembers a similar moment: standing over a flask, thermometer in hand, checking crystalline formation while the whole synthesis hinges on that transition from solid to liquid.
The melting point of 9-anthraldehyde isn't just a number for a technical data sheet—it signals purity and reliability. An impure sample will melt irregularly, and that tells you something’s off. This clarity makes it easier to weed out mistakes early, rather than hunting for impurities downstream in complicated reaction mixtures. Academic studies, such as those from the Journal of Organic Chemistry, keep pointing out how key a sharp melting point is for quality control. If you’re scaling up for material science or pharmaceuticals, those few degrees keep processes reproducible and safe.
There's another side to this temperature. Those manufacturing organic semiconductors or bright yellow dyes use 9-anthraldehyde for more than its structure—the melting point determines how smoothly a process will run. Too low, and operations in summer labs turn sticky. Too high, and you waste energy and risk damaging equipment. Efficiency depends on getting that number right. Energy costs were a big topic at my previous workplace, and letting a hotplate run all day for an extra ten degrees caused unnecessary delays and costs. Supervisors pay attention when something starts costing more than it should per batch.
My experience echoes what regulatory chemists follow—for health and safety, volatile organic compounds need close monitoring. The melting point links to vapor pressure, exposure risks, and even waste handling. Neglected, it can nudge a working day toward hazard. Occupational safety reports highlight cases where a few overlooked degrees led to cleanup headaches or exposure breaches. Proper documentation and clear communication about values like this save both time and health.
The solution isn’t glamorous, but it works. Every institution should keep a verified copy of physical data for target molecules. Double-checking values in peer-reviewed sources like the Merck Index or reliable chemical databases helps prevent mistakes from multiplying. Training new lab staff to notice a melting point discrepancy as an early warning sign keeps quality high. Calibrating equipment every few months used to feel tedious, but it saved us from disaster more than once.
Accuracy about details such as the melting point for molecules like 9-anthraldehyde doesn’t just polish up a research report. It forms the backbone of work that aims higher: safer workplaces, leaner production, and stronger trust between industry and science. As data-sharing becomes more important across fields, something as humble as a melting point starts to look like the cornerstone that it really is.
In chemistry work, little things add up. I learned this from long hours in graduate labs, where a single slip-up with storage turned an ordinary weekday into a hectic clean-up. Chemicals like 9-Anthraldehyde are useful for organic synthesis and research, but their quirks demand respect—starting with how they're put away at the end of the day.
At room temperature, many aromatics drift by just fine, but 9-Anthraldehyde likes things stable. Heat and sunlight can do a number on its structure, causing degradation. I once watched an old bottle, left too close to a window, develop odd tints over months—a sign something hadn’t stayed pure. Fluctuations in climate can change outcomes in the lab, especially where precise yields or spectral purity matter. So, chemistry veterans stash these bottles away from direct sun, ideally in a cool, dry cabinet—a place where temperature swings won’t tamper with the compound’s integrity.
Some compounds live easy around water. 9-Anthraldehyde isn’t one of them. Exposure to humidity can prompt unwanted side reactions or stubborn clumps. After spending too many hours chipping crystals out of ruined bottles, I now reach for a sealed, airtight container every time. Silica gel packets aren’t just for packaging shoes—they make sense in chemical storage too. You drop a fresh one in, keep the lid tight, and you save yourself a headache later.
People new to the lab sometimes underestimate glassware. Any old jar won’t do. Proper screw caps, glass-stoppered bottles, or amber glass work best for sensitive chemicals. Plastic leaches, clear bottles let in UV rays, and cheap stoppers never seem to fit snugly. I’ve seen labeled flasks with cracked tops corrode from exposure. Paying attention to the container itself isn’t about being picky. It’s about keeping samples usable—and safe—over time.
It might sound trivial, but a clear label with the chemical name, date received, and even a hazard notice sets good labs apart from bad ones. I’ve walked into storage rooms where faded labels caused confusion about what’s inside. For something like 9-Anthraldehyde, which isn’t exactly a household name, good labelling cuts down the chance of mix-ups, mistaken uses, or ugly surprises.
I spent some years in a teaching lab where open shelves were common—until someone dropped a strong oxidizer next to a shelf of organics and a brief scare followed. Chemicals with reactive tendencies, even ones that seem benign, should have space away from incompatible reagents and strong oxidizers. Storing them in ventilated cabinets, not next to acids or bases, wasn’t just a rule on the wall; it was how we kept the place safe.
Proper storage depends on more than a list taped to the door. In places where staff received regular updates on chemical handling, fewer accidents and less waste cropped up. Training didn’t just cover what’s hazardous—it made respect for safe storage routine. From undergrad labs to industry shops, good habits around 9-Anthraldehyde and similar reagents protected people and research projects.
If you treat every bottle like you want clean results and a safe lab, you do right by your future self—and everyone around you.